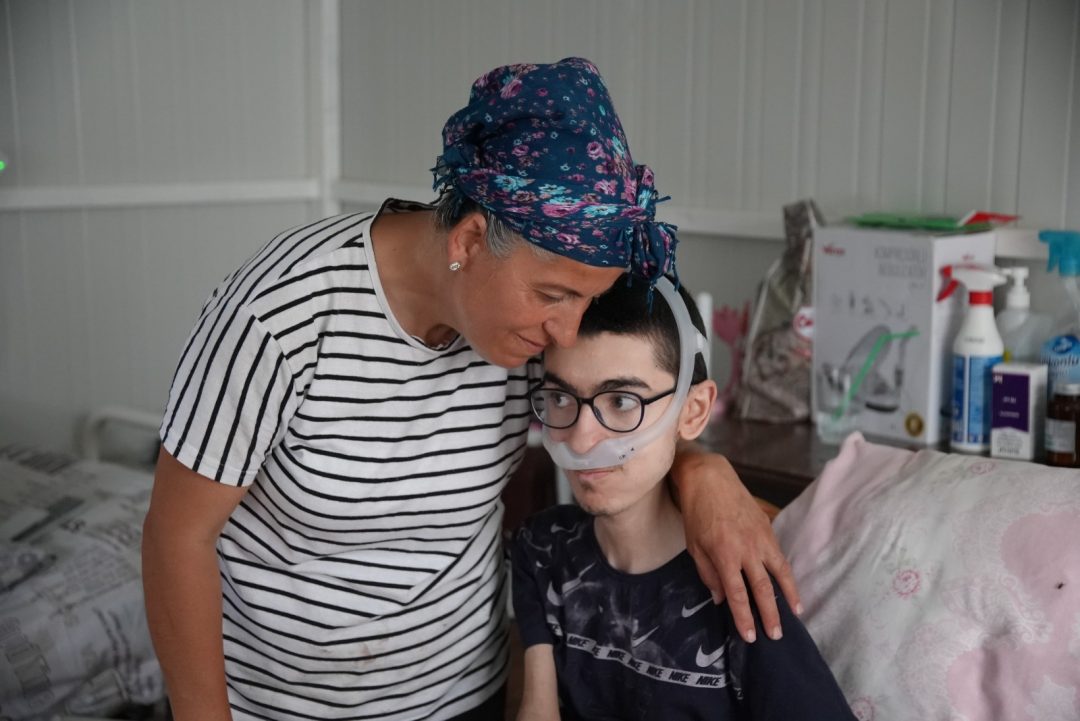Summer is 6 years old and we live in Australia.We found out Summer is a carrier for Duchenne Muscular Dystrophy when she was 5yrs old. She was very late to sit crawl and walk. She finds it difficult to run and complains a lot of tired legs. I knew there was something different about Summer her whole life, she has had elevated CK levels from 2 yrs old and was finally diagnosed as a carrier of DMD after a full chromosomal microarray was done. I as her mother was tested also am not a carrier.Melissa Brunet
Story by Dimitrios Athanassiou
Hi my name is Dimitris I have two beautiful boys Hermes and Hector with my wife Omaira and we live in Athens, Greece. My older son Hermes has DMD.
What is your dream? My dream is my son’s generation to be the one that in their lifetime we eradicate Duchenne.
What do you like to do? When I find some free time, which isn’t very often, I like to take my family to the beach.
If you could change something in the world, what would you change? I would change the future of the rare diseases.
If you could invite any three people dead or alive to dinner who would that be? the ones that will find the cure for DMD
What was your favorite experience? From going down the Amazon on a boat to riding a bike at 300 kph, nothing was more exciting for me than being present at the births my two children.
What is your best memory? Their first smiles
What is your funniest memory? The expression on the face of my first son when he found out that he is not alone! My typical day starts s at 7am when I wake up and get Hermes ready for nursery before dropping him off. Then it’s straight into my email account where I will exchange messages, build contacts, talk to parent, discuss new information and trials, upcoming medical conferences and parent advocacy. I will take around 30-40 phone calls day from at home and overseas on a typical day for this purpose, then pick up my son around 3.30pm, more emails, more campaigning, stop for dinner, maybe take a final phone call or join a conference call around 10pm, then asleep at 2 or 3 am. If all the above happen I consider it a good day. The next day I do it all over again happily!
What will you do tomorrow? The same as what I will do today, but with a small change because we are driving three hours to jump on a boat and have a this weekend in Cyclades.
Who is your favorite person to have around you? My wife because she beautiful, kind, patient ( with me ) and very smart (says Omi ? )who is checking what I am writing !!!!)
My day is a success when….my family is smiling and we are one step closer to cure DMD
Story by Andrew Sedmihradsky
Starting on Father’s Day this year, my four year old son Max and I set off on a 600 km cargo bicycle ride from Ottawa to Hamilton along the Trans Canada Trail – a journey we named ‘Max’s Big Ride’. Max and I made this ride to advance awareness about Duchenne muscular dystrophy and also to raise money for research. So far the ride has raised close to $55,000.
Max’s Big Ride was a true family affair as Max’s grandparents and mother also hit the road with us in support roles. It was a tremendous adventure and we saw lots of wildlife along our scenic route and met even greater numbers of kind and incredible people. Max and our family were welcomed warmly by cheering crowds at various points along the way and we were all deeply touched and incredibly thankful for all the support we received.
I am happy to report we made it to the finish line, arriving in Hamilton’s Bayfront Park on Canada Day, July 1st, to the cheers of our friends and supporters. While Max’s diagnosis has been very hard for our family to deal with, we have decided to roll up our sleeves and fight this horrible disease as best we can. This year’s ride is just the beginning, and planning for our 2016 fundraiser will begin in a few weeks.
Story by Maria & Hanna Fries
My name is Maria Fries-Lindgren. I was born in Sweden and I also went to schoolthere. 25 years ago I moved to Switzerland to work and I’m still here. I’m marriedwith Daniel and we have two lovely children. Mattias, 11 years old who hasDuchenne, and Hanna, 9 years old. I do not have a professional life, I stopped working when my son was born. I like very much being mom and housewife. I also like writing, reading, cooking, gardening and to jog. I do some voluntary service with the websitewww.duchenne-schweiz.ch.I answer mails from families recently diagnosed with duchenne and I try to help andsupport them. I was recently elected as a board member in the Swiss muscle society(Schweizerische Muskelgesellschaft).
My dream, my biggest dream, is that a cure for Duchenne soon will be found.During the weekends I like to do something with my family. Bicycling, walking,playing games…If I could change something in the world, I would end all wars.I would like to invite the pope, Wladimir Putin and my grandmother, who died when Iwas only one year old. My favourite experience is the birth of our two children.My funniest memory is when I meet my mother (once or twice a year) and we sittogether and talk. Suddenly we start laughing, just like that. A glance, and weexplode in a warm hearted laugh, without a reason. It happens every time we meet.I wake up at 6. Then I get up, make lunch for my husband and prepare breakfast forthe children. After they are gone to work and school, I go out to jog. After that I dosome washing and cleaning. Some days I have to do some office job, answering e-mails or preparing some papers. At 11 I cook lunch for the children. We eat at 12.They go to school again at 13.15. Mostly I work a little bit again. Kids are back at15.15. I help them with their homeworks. My son likes to bake, cakes and bread,and we often do that together. I prepare dinner and when my husband comes home around 18.30 we eat. The children go to bed around 20. Then my husband and I sit on the sofa with a cup oftea and talk a little bit. Some evenings he has to work a little bit on the PC, then Ijoin him on my PC. Or I read a book. I go to bed around 23.
Yesterday I met 10 great duchennemoms! I had organized and invited duchennemoms from the german speaking part of Switzerland to meet and exchangeexperiences, and to have a great day together, of course. Some of them I met forthe first time. I was overwhelming to learn about so much power and strength andlove! We took a boat trip to a small place where we got off the boat and had a nicelunch in a beautiful restaurant. We talked and talked. We laughed and we cried. Lateafternoon we went back with the boat, an old steamer, and enjoyed the sun, thewind and the beautiful sight of the mountains. It was a wonderful day!Tomorrow I will go for a jog first thing in the morning. Then I will iron a load ofclothes. I will have to pack a small suitcase also.Since two years now, my son takes part in a clinical trial in Paris and we travel every week with him to France. We take the train (TGV) from Basel. During travelling I will do some work on my laptop, answering e-mails and preparing some documents. IfI get tired I listen to music and close my eyes. When we arrive in Paris I walk withmy son in the wheelchair to the hotel, it’s a 15 minutes walk along the river Seine. After our arrival in the hotel we have a procedure, it’s always the same… We both sitin the bed, my son watches TV and we eat a sandwich. During a commercial break hetakes a short shower, during the next one he brushes his teeth. At 22 we switchof the TV and the light. Holding his sweet little hand we fall asleep. We would bothprefer to be at home in our own beds, but we know how important the clinical studyis and as he never complains about it, I neither do I so.My favourite person to have around me is… I don’t know.
I don’t have any favouritepersons to have around me, except my family. I like be being alone, so maybe theanswer is: nobody ?I’m Hanna Fries, I’m the sister of Mattias. Mattias has Duchenne. I’m 9 yearsold. I live in Switzerland. I would like to be a popstar.I like to draw, play with barbies and Legofriends.I also like to play outside. And of course I love to sing and to dance.Everything should be for free! And that everybody in the world has fresh andclean water. I would like to invite Taylor Swift, my grandfatherand my best friend Lorena.My favourite experience was to learn to dance.My best memory is from a camp together with other kids.My funniest memory is when my little cousin went with us to an amusementpark. There she went with a ghost train. I was waiting outside. When she came out she laughed so much that she cried and I laughed with her. I wake up at 6.30. After I have got dressed, I drawa little. Then I have breakfast.
I go to the toilet and brush my teeth.At 7.50 I go to school with my brother. School starts at 8.10. At 9.40 there is a break where I play outside with my friends. Schoolstops at 11.40. Then I go home and have lunch which my mother cooked. Some days I have school in the afternoon. Then I leave at 13.10 and come back twohours later. I have homework everyday to do. Sometimes I play with a friend, or go to a dancelesson. I have dinner at 18.30, then I brush my teeth. I go to bed at 20.30.Yesterday I went to my grandmother and had lunch with her. In the afternoonwe took a boat trip on the lake of Lucerne. My mother picked me up in the evening and we went home.Tomorrow I will have to go to school again. I haveno choice!My favourite person is my best friend Lorena.My day is a success when… nothing bad happened to me and nobody saidsomething stupid to me.
Breaking Barriers: The Story of Bert Gooris
Meet Bert Gooris, a 32-year-old from Belgium, living with Duchenne muscular dystrophy. This year, for World Duchenne Awareness Day (WDAD), he is one of our Duchenne Ambassadors. Over the next few weeks, Bert will be sharing his journey on breaking barriers in his personal life.
About Bert Gooris
“Hello, I’m Bert Gooris, a 32-year-old living with Duchenne muscular dystrophy in Leuven. Though I reside with my parents, I have my own space in the house, complete with a desk, room, and bathroom. I am a positive person and a dedicated family man. My 21-year-old sister also lives with us, and my older brother, who doesn’t have Duchenne, resides in Landen. It was a shock for me and my family to learn that my sister is a carrier of Duchenne muscular dystrophy, when she was 18 years old. Recently, I got a tattoo symbolizing the strong bond between me and my siblings. My parents have always supported my participation in various activities, like playing soccer and riding my bike. Although I faced bullying in school, I stood up for myself and continued my education smoothly, making some of the best memories during my college years.”
Daily Life with Duchenne
“Due to Duchenne, I had to readjust my ideas about what I wanted to become in the future. At home, when we talked about further studies, I wanted to attend culinary school to become a chef. However, it soon became clear that this would not be feasible in the long run due to the loss of function in my arm and leg. That’s why I decided to pursue something administrative because working with computers would remain possible for a longer time. Even in football, I had to admit that playing matches became too difficult, and eventually, I couldn’t participate in training sessions anymore, so I wouldn’t become a footballer.”
Duchenne Ambassador
“At school, I had to stand up for myself, explain that I had the disease, explain why I couldn’t run as fast, and why I was heavier than others. This made me a person who cannot tolerate injustice and will stand up for the weaker ones in society. As I grew older, I encountered more stories of injustice or shortcomings, and I became more aware that many people still don’t know about the disease despite the efforts made. That’s why I see it as my duty, as an adult living with Duchenne, to take on the role of ambassador now. I want to share my story with other individuals with Duchenne to encourage and support them. By doing so, I aim to create global awareness.”
Interpreting ‘Breaking Barriers’
“Well, it’s about time we dare to break those barriers and push for change. Change in inclusion, belonging, being informed about medication, assistive devices, the right to certain benefits, and the steps to follow during important milestones in life. It should be clear for everyone what rights they have and where to access relevant information. The time has come for people with disabilities to be fully accepted by society, and administrative processes for them should be significantly streamlined.”
Key Message
“It’s still a tough world for people with disabilities. We are just like any other individuals with the right to be fully included, 100%. We lead lives just like everyone else. If we collaborate and are truly heard, we can overcome many obstacles together.”
Future Plans
“In the future, everyone should be aware of these diseases. Efforts should be made to grant certain rights to people living with Duchenne and Becker, or any other conditions.”
We invite you to follow his journey by following him on social media.
- Facebook: Bert.Gooris
- Twitter: @UmbertosMission
- Instagram @Umbertos_Journey
- LinkedIn: Bert Gooris
Breaking Barriers: The Story of Brad Miller
In this interview, we meet Brad Miller (47), a Canadian blogger living with Becker muscular dystrophy (BMD). Brad shares his remarkable journey of adapting to challenges posed by the rare disease, and pursuing his passions with confidence and determination. He talks about his childhood experiences to creating his blog, “My Beckers Story,” and founding “Laps for Muscular Dystrophy.” Brad’s story is an inspiring testament to breaking barriers and raising awareness for BMD.
About Brad Miller
“My name is Brad and I’m a Canadian blogger living in Ontario with Becker muscular dystrophy. As a child I was having trouble keeping up with the other kids my age. This led to a visit to a family doctor who thankfully had some knowledge about muscular dystrophy. At the time, my enlarged calf muscles grabbed his attention. So, at the age of 10 I was sent off for some testing which also included me having a muscle biopsy. After that my parents were given the news that I have Becker muscular dystrophy.”
Becker muscular dystrophy and daily life
“As a child with Becker muscular dystrophy, I could still enjoy riding my bike and playing with friends, but certain activities like jumping, climbing trees, or participating in running sports were challenging for me. However, I didn’t let these limitations bother me and learned to adapt at a young age, thoroughly enjoying my childhood. The real challenges surfaced in my late teens, experiencing frequent falls and needing several days to recover after pushing myself too far physically, especially during trips to amusement parks. Nowadays, I do my best to adapt to the challenges that come my way, as walking becomes more difficult. I’ve started using a mobility scooter for longer distances and a cane when required. Adapting to my changing needs is an ongoing part of my life with Becker muscular dystrophy.
Following dreams with Becker MD
Having this condition never stopped me from following my dreams. Throughout my life, I’ve pursued my passions, including writing and performing my songs. I’m proud to have published two articles online, featuring my list of the Top 10 vehicles for people with disabilities and reviewing a newly accessible vehicle. Unfortunately, a few years ago, I suffered a work-related injury, leading me to stop working. However, this didn’t deter me; instead, it inspired me to create the My Beckers Story blog, where I share my experiences of living with Becker muscular dystrophy. A few years later, I founded Laps for muscular dystrophy, where racing teams contribute to raising awareness and funds for muscular dystrophy charities. My journey has been about adapting and persisting, pursuing my passions and advocating for others in similar situations.”
My goal for WDAD
“For WDAD in 2023, it is my passion for raising awareness and understanding of the issues we face. As I really believe when people better understand our daily lives it can result in more support of our community. With the end result being more support of muscular dystrophy charities and of the important research & studies currently taking place. After all, the hope of many is seeing new treatments and the possibility of one day finding a cure.”
About the Breaking Barriers Theme
“To me, this year’s WDAD theme represents the determination of our community to support each other by working together to advance the cause of raising much-needed awareness and funds to help improve the lives of current and future generations. I think raising awareness is a great way of breaking barriers by making more people aware of the unique challenges we face. This resulting in more support and understanding.”
Why I’m joining as Becker Ambassador
The main reason I have decided to share my story is to show others they are not alone. To also show people that perseverance in life is so important. I’ve certainly faced many challenges in life. Some related to living with Becker Muscular Dystrophy others just the regular challenges of life. In my case I survived being bullied as a child as unfortunately the other children would make fun of the way I walk, call me names and once even spit on me. It was easy but I survived and still no child living with any form of Muscular Dystrophy should have to deal with being bullied. So, another hope of mine in raising awareness of BMD is that it makes more children aware of those living with MD so that future generations don’t have to deal with bullying. Another hope of mine is that reading my story helps parents better understand what it is like to live with BMD to hopefully show people we can live our lives to the fullest, have careers, start families and enjoy life.
Since the start of sharing my story, I’ve shared it openly and honestly by not shying away from addressing any issue affecting those with disabilities. Simply because I believe we can learn a lot from each other’s stories. That is why I often encourage others to start sharing their stories as it certainly can make a huge difference in people’s lives knowing they are not alone in their experiences. In a way letting them know we are all in this together with the same hope of one day seeing a cure for Muscular Dystrophy.”
Key Message
“So, let’s keep sharing our stories and raising awareness. Supporting WDAD is key in encouraging everyone. Even those outside our community to join us in the fight against Muscular Dystrophy.”
We are inviting you to follow Brad with his Becker story through his website and social media channels:
- Website: mybeckersstory.blogspot.com
- Instagram: @mybeckersstory
- Twitter: @mybeckersstory
- Facebook: facebook.com/mybeckersstory
Breaking Barriers: The story of Anil Jalhan
Meet Anil Jalhan, also known as “The DMD Guy” on social media. At 26 years old, he shares his journey with Duchenne muscular dystrophy (DMD). Despite facing physical challenges, Anil broke barriers by graduating from university and become an independent individual. As a Duchenne Ambassador, he aims to create a positive community, raise awareness, and inspire others to share their stories.
About Anil Jalhan
“My name is Anil and I am 26 years old, living in the UK. I thought I would talk about my Duchenne Muscular Dystrophy (DMD) journey. Prior to my diagnosis I grew up not knowing I had it and even my parents didn’t know. As I was the first boy born after my parents had two healthy children, it was likely I would be healthy too. Eventually we found out that boys were mostly affected and how it can be passed down in the family.
I don’t remember much, but I remember walking to nursery and school with my mother. Eventually my mother would receive a phone call from the school informing her that I kept falling frequently and was moving much slower than other kids. This was the very first signs of the condition. Once we decided to get me checked out, it was clear what was wrong with me. I was diagnosed with Duchenne Muscular Dystrophy (DMD)
After the confirmed diagnosis, my future was full of questions and uncertainty, as I didn’t know how it would affect me. I would continue to move forward while going through these physical changes. I would experience being wheelchair bound and weak whilst enjoying my primary school days.
Once the time was right, I would later make my transition to my new special needs school. This was a huge moment of my life, as the school helped me become who I am today. Giving me the confidence to become an independent disabled person.
I am the person today because of this experience. I believe my disability has taught me many valuable life lessons and has helped shape the person I am today. My journey has been difficult with many barriers and many still to come. But I aim to just keep going. I usually like to say my future may not be clear, I may have a disability, but this will not stop me from achieving my dreams.”
Daily life with Duchenne
“I want to say being physically weak and all the constant changes I have and will go through. But I did come to the realization that it’s something I had to just accept and I could still pursue my dreams. One of my dreams was to graduate from University and I did back in 2019. With the help of assistive technology this was possible. One major barrier was my housing situation which I will talk about in another post hopefully. But this really was a serious one which stopped me from getting out of the house and sometimes even going to university.”
Duchenne Ambassador
“With Duchenne and my page, my goal has always been to create a positive Duchenne community for other DMD guys like myself. Also to raise as much awareness as possible. The WDAD has a similar goal which really motivated me to get more involved. I also wanted to share my story to hopefully encourage others to share theirs.”
Interpreting ‘Breaking Barriers’
“As someone living with Duchenne Muscular Dystrophy (DMD) like many we go through many life changing moments during our Duchenne journey. With many battles and emotional moments. From not being able to walk or losing the ability to play your favorite sport. I think my interpretation of ‘breaking barriers’ is to have the ability to accept these changes and adapt to them while trying to remain positive. Lastly, I also think going through all these trials and tribulations and still being able to achieve something is breaking barriers. No matter how big or small the goal is.”
Key message
“The journey can be quite emotional at times and tough. The only thing we can do is try our best to enjoy life no matter how long it is. Also to create these precious memories. Through my stories I hope to show the reality of Duchenne but also try and show how life could be enjoyed as well. Another general message to the world is that everyone is affected by this, men, women and kids so let’s do something about it.”
Future goals
“I hope to see more people with Duchenne continue to break barriers and to share their story. Further increasing the awareness for Duchenne and Becker Muscular Dystrophy. In terms of my future I also will continue to overcome challenges. I hope to make use of any opportunities I get to raise awareness for Duchenne. Lastly I remain hopeful that there will be a cure in the future mainly because of our awareness efforts.”
We invite you to follow Anil’s story via Instagram and TikTok. Here he shares messages on resilience and the pursuit of joy in life. All while advocating for a future cure through increased awareness efforts. We are looking forward hearing your story about breaking barriers!
Instagram & TikTok: thedmdguy
Together, we are stronger
Today, the global awareness raising campaign for Duchenne and Becker muscular dsytrophy has launched.
Breaking Barriers: WDAD 2023 Documentary Set to Launch
On September 7, 2023, the World Duchenne Organization will be launching an impactful documentary titled ‘Duchenne: Breaking Barriers’. This will be done to mark World Duchenne Awareness Day (WDAD) 2023.
Directed by Nicoletta Madia, this documentary promises to be a powerful tribute to the lives of individuals living with Duchenne and Becker Muscular Dystrophy (DMD/BMD) around the world. The WDAD Documentary ‘Breaking Barriers’ fosters global awareness and understanding of dystrophinopathies through the power of storytelling.
WDAD 2023 Documentary
The theme ‘Duchenne: Breaking Barriers‘ aims to shed light on the remarkable journeys of people living with this rare disease, showcasing how they are each breaking barriers in their personal lives. One of the unique aspects of this documentary is that it is collecting experiences from different parts of the world, offering a global perspective on the struggles and triumphs faced by individuals with DMD/BMD.
The film takes viewers on a journey through various countries, including Turkey, Mexico, and Italy. Nicoletta Madia and her team have selected inspiring stories of courage and resilience. By sharing the experiences of people from diverse cultural backgrounds, the documentary highlights the different types of barriers people with Duchenne face and the innovative ways they challenge these obstacles.
Upon its launch on September 7, we urge everyone to share the Breaking Barriers WDAD 2023 Documentary. By doing so, we can amplify the voices of those living with DMD/BMD. Additionally, we can advocate for greater support, research, and resources to address their needs effectively.
Breaking Barriers
World Duchenne Awareness Day serves as a reminder that together, we can make a difference in the lives of those living with Duchenne and Becker MD. As the launch date approaches, please mark your calendars and make plans to share “Duchenne: Breaking Barriers” with your friends, family, and colleagues.
Stay tuned for the release of the Breaking Barriers WDAD 2023 Documentary on September 7. Join us in spreading the message with the world. Together, we can make an impact and create a better and more supportive world for people living with DMD/BMD.
Breaking Barriers: The story of Troy Chauppetta
Meet Troy Chauppetta, a 28-year-old Duchenne Ambassador from Massachusetts. He’s a USA wheelchair Power soccer team player, graphic/website designer, and living independently. Troy defies odds with his hand-controlled vehicle, roommates, and dog. He has a strong message: Disability is ability. He’s breaking barriers and advancing Duchenne awareness through technology, support, and determination.
About Troy Chauppetta
“My name is Troy Chauppetta, a 28-year-old from Massachusetts, USA. I’m passionate about sports, playing for the USA wheelchair Power soccer team. I am all about independence, driving my vehicle with hand controls and living with roommates. I’m also a proud dog owner. When I’m not on the field or exploring with my dog, I channel my creativity as a Graphic Designer and website designer.”
Daily life with Duchenne
“My daily life with Duchenne is great defining all the odds. My Personal Care Assistants get me up and ready to take on the day. Most of my creative work is done on the computer. I take care of my apartment and make sure my dog is all set for the day. But as people know, living with a disability there are lots of ups and downs but I try to keep it positive. I have a twin brother who also has Duchenne and having each other helps me with the daily life living with Duchenne. Because I have someone always by my side that I relate to!
Duchenne Ambassador
“My motivation stems from a strong desire to share my journey, demonstrating that one can overcome challenges while living with a disability. I’m determined to inspire and show that defying the odds is possible.”
Interpreting ‘Breaking Barriers’
“To me, ‘Breaking Barriers’ signifies the ability to lead a fulfilling life despite a disability, achieving anything by harnessing determination and willpower. You can live a great life with a disability and do with you put your mind to.”
Key message
“My message is simple yet powerful: I want people living with Duchenne and Becker muscular dystrophy not to limit themselves just because they have a disability. I see the world Ability In disability you have the Ability to do with you dream.”
Future goals
Troy is positive when asked about the future in terms of breaking barriers and advancing awareness for Duchenne/Becker muscular dystrophy. “I see lots of wonderful things happening in the future with Advancing awareness with all the technology and advancements and the wonderful support groups we have!”
Looking forward, Troy is optimistic about the future, anticipating significant progress in awareness and support for Duchenne and Becker muscular dystrophy through the integration of technology and the unwavering strength of support networks.
We invite you to follow Troy Chauppetta’s inspiring story via social media. Inspire others to break down barriers in Duchenne or Becker by sharing your story.
Instagram: @_tc09t
Facebook: Troy Chauppetta